For children with epilepsy, developmental delay, or intellectual disability, the diagnostic journey is often long, complex, and emotionally exhausting. It is also expensive for health systems and payors, frequently involving repeated hospitalizations, emergency visits, and a series of inconclusive tests—without delivering the answers families and clinicians need.
New research presented at the American College of Medical Genetics and Genomics 2026 Annual Meeting demonstrates that genomic testing can fundamentally change that trajectory. The Genetic Sequencing Analysis has Value in Economic Savings (SAVES)-Kids Study, a real-world Health Economics and Outcomes Research (HEOR) analysis led by GeneDx, demonstrates that our ExomeDx™ and GenomeDx™ significantly reduce total healthcare costs in the 12- and 24-months following testing for both Medicaid and commercially insured children.
- Savings were primarily driven by fewer hospitalizations and reduced emergency room utilization – patterns consistent with accurate diagnoses and more targeted, high-value care after genomic testing.
- The study found that all test result types (positive, possible, and negative) offer significant average cost savings and improved health outcomes.
The findings make clear just how transformative early genomic testing can be. A genomic answer—positive, negative, or uncertain—reshapes the clinical journey and reduces avoidable, high-cost care. By delivering answers sooner, exome and genome sequencing guide more precise care, improve outcomes for children, and significantly reduce healthcare use and costs.
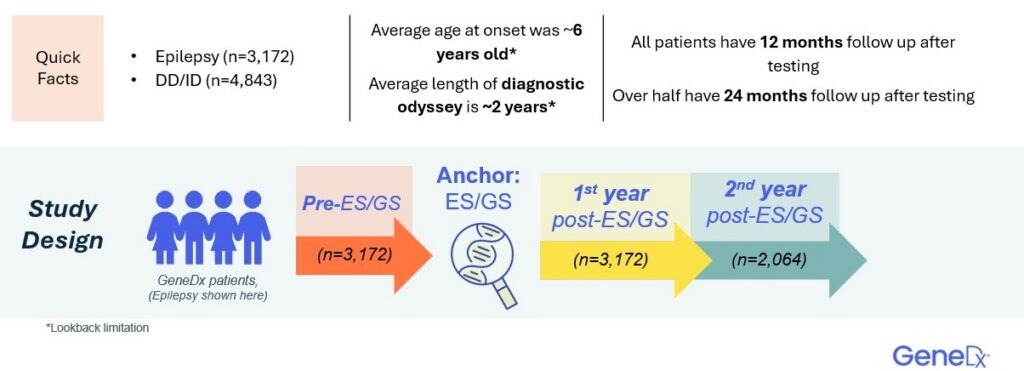
Study overview
The SAVES Kids study is a large, real world, retrospective cohort study linking deidentified GeneDx testing results with Komodo Health claims data. It includes 8,015 pediatric patients, two thirds of whom were covered by Medicaid, reflecting a diverse and medically complex population.
Genomic Testing Reduces Healthcare Costs and Improves Outcomes
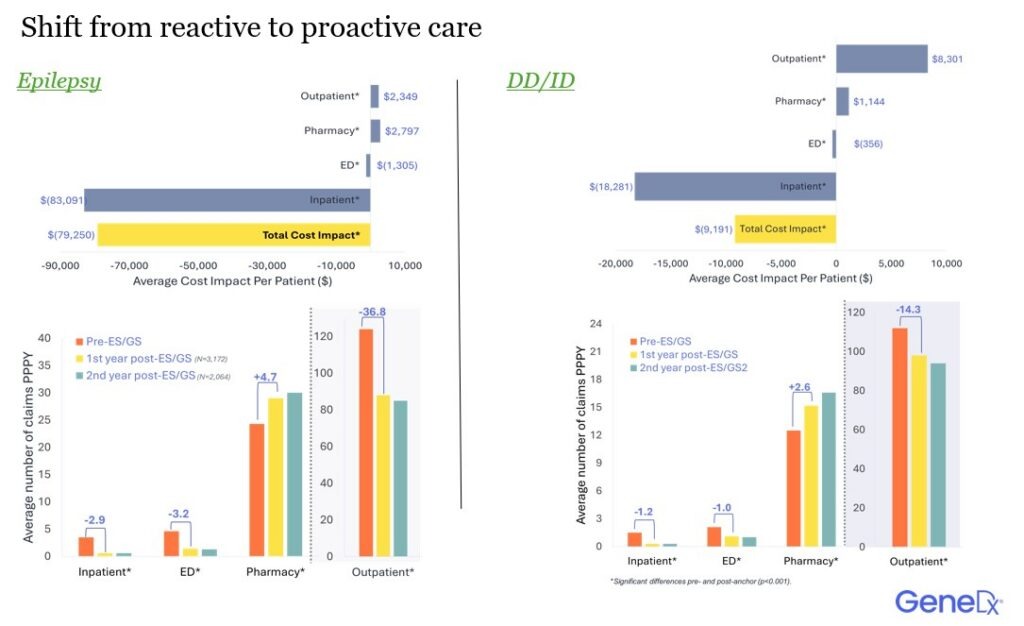
Across both epilepsy and DD/ID cohorts, ES/GS testing led to significant reductions in acute care use, including fewer hospitalizations and emergency room visits. These improvements occurred regardless of test result type, highlighting that genomic testing provides clinical value beyond establishing a diagnosis.
Key highlights:
- 61% reduction in healthcare costs for children with epilepsy in the 12 months after ES/GS, saving an average of $80,000 per child.
- 79% fewer hospitalizations and 48% fewer ER visits for children with DD/ID in the year after testing.
- ES/GS shifted care away from highcost acute interventions toward proactive, targeted management.
- Medicaid and commercially insured children saw similar trends, underscoring broad applicability and equity in impact.
- These findings reinforce that ES/GS should be a first-tier test, especially when neurological symptoms, such as seizures, begin.
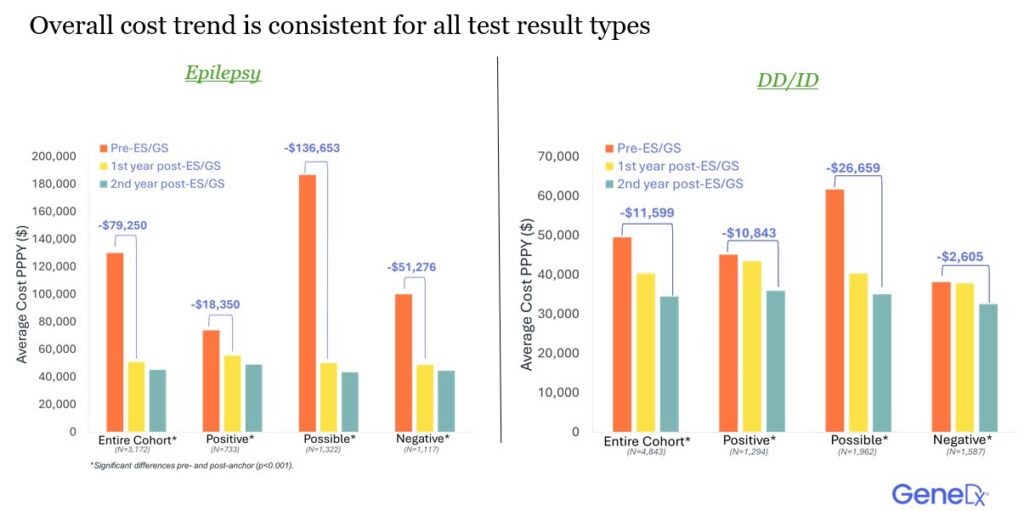
Epilepsy: Significant Cost Savings and Fewer Acute Care Event
For children presenting with epilepsy, ES/GS dramatically changed the care trajectory:
- Average total healthcare costs dropped from $130,048 PPPY to $50,798 PPPY, a 61% decrease.
- Hospitalizations fell from 3.5 per year to 0.6, and ER visits dropped from 4.6 to 1.4 per year.
- Total healthcare interactions declined by 32%.
- Outpatient claims decreased even as outpatient and pharmacy costs rose slightly, reflecting more targeted management, not more utilization.
- Combined with findings from GeneDx’s 2025 seizure dataset, the evidence is clear: stepwise testing delays diagnosis and adds avoidable costs, while ES/GS identifies twice as many diagnostic findings as panel tests.
DD/ID: Substantial Reductions in Hospitalization and Acute Care
For children with developmental delay or intellectual disability:
- Total costs dropped by $9,191 PPPY in the first 12 months after ES/GS, and $11,599 PPPY by 24 months post testing.
- Hospitalizations fell by 79%, the largest contributor to savings.
- Emergency visits declined by 48%.
- Outpatient claims decreased by 13%, even as outpatient/pharmacy costs increased—again pointing to more proactive, effective care.
- This pattern across both cohorts demonstrates the broad clinical efficiency created by genomic testing.
A 1.8 Year Gap Between First Symptoms and Testing
Despite strong clinical guidelines, children with neurological conditions often wait nearly two years from their first ICD10 code for epilepsy or DD/ID before receiving ES/GS. Many also had 3–4 additional medical conditions, illustrating their complexity and the need for broad, comprehensive testing early in the journey.
What This Means for Clinicians, Payors, and Families
The SAVES Kids findings demonstrate that ES/GS is not just diagnostic, it’s transformative. By reducing acute care, guiding more precise treatment, and providing clarity at every result type, genomic testing leads to:
- Better health outcomes
- More efficient resource use
- Substantial cost savings
- Reduced parental uncertainty
- More equitable access across insurance types, including expanding Medicaid coverage for genomic testing
For epilepsy and DD/ID, conditions marked by complexity and urgency, genomic testing belongs at the beginning of the diagnostic pathway, not the end.
Why Earlier Testing Matters
Despite clinical guidelines recommending exome and genome sequencing as first-tier tests for many pediatric neurological conditions, testing is still frequently ordered late in the diagnostic journey.
The SAVES-Kids study adds compelling real-world evidence that earlier genomic testing can accelerate the transition from reactive, high-cost acute care to more efficient and targeted treatment pathways, aligning coverage policies with guidelines to accelerate diagnosis while reducing avoidable healthcare utilization.
“Exome and genome sequencing deliver better health outcomes and measurable cost savings. The standard of care must reflect that reality,” said Linda Genen, MD, MPH, FAAP. “The earlier genomic testing is implemented, the faster children receive targeted management, and the more efficiently health systems and payors can allocate resources. The data is clear—genomic precision medicine is not just better care, it is smarter healthcare.”
“Exome and genome sequencing deliver better health outcomes and measurable cost savings. The standard of care must reflect that reality,” said Linda Genen, MD, MPH, FAAP. “The earlier genomic testing is implemented, the faster children receive targeted management, and the more efficiently health systems and payors can allocate resources. The data is clear—genomic precision medicine is not just better care, it is smarter healthcare.”
Building a Smarter Healthcare System
As healthcare systems increasingly prioritize value-based care, evidence demonstrating both improved outcomes and reduced healthcare utilization is critical.
The SAVES-Kids study adds to the growing body of real-world evidence showing that genomic testing can help health systems move from reactive, high-cost care to more proactive and targeted management — delivering better outcomes for patients while supporting more sustainable healthcare spending.
These findings build on GeneDx’s mission to transform rare disease diagnosis through genomic data. As the leader in rare disease diagnostics, GeneDx has delivered more rare disease diagnoses than any other organization worldwide.
This impact is powered by GeneDx Infinity™, the world’s largest rare disease dataset—comprising more than one million exomes and genomes and more than 2.5 million tests. By combining these insights with real-world healthcare data from Komodo Health’s Healthcare Map®, GeneDx continues to generate evidence that advances precision medicine.
Together, these data demonstrate something increasingly clear across the healthcare system: genomic testing doesn’t just provide answers for families—it creates a more efficient and sustainable model of care.
And for children with complex neurological conditions, that shift can change everything.




